By Veranova

![]() Antibody-drug conjugates (ADCs) are a rapidly developing therapeutic modality within the pharmaceutical industry. This article will explore approaches to unlocking the potential of these molecules, including how CDMOs can leverage technical expertise to develop ADCs with confidence.
Antibody-drug conjugates (ADCs) are a rapidly developing therapeutic modality within the pharmaceutical industry. This article will explore approaches to unlocking the potential of these molecules, including how CDMOs can leverage technical expertise to develop ADCs with confidence.
ADCs: A Fast-Growing Therapeutic Area
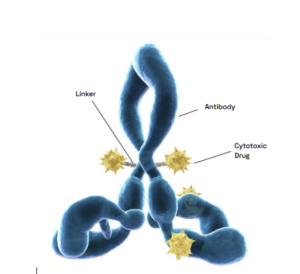
 Antibody-drug conjugates (ADCs) are an innovative, emerging therapy area. Primarily used in oncology, ADCs are comprised of a monoclonal antibody, a cytotoxic drug payload, and a chemical linker. The action of an ADC depends upon the qualities of each of these components. By combining the specificity of monoclonal antibodies with the potency of cytotoxic small molecules, ADCs facilitate stronger clinical responses and fewer systemic side effects for patients.
Antibody-drug conjugates (ADCs) are an innovative, emerging therapy area. Primarily used in oncology, ADCs are comprised of a monoclonal antibody, a cytotoxic drug payload, and a chemical linker. The action of an ADC depends upon the qualities of each of these components. By combining the specificity of monoclonal antibodies with the potency of cytotoxic small molecules, ADCs facilitate stronger clinical responses and fewer systemic side effects for patients.
With the global ADC market predicted to reach USD 24.01 billion by 2030, the race is on for developers.1 Currently, around 13 ADCs are commercially available, and numerous formats are further being explored.2 However, despite significant breakthroughs in ADC development, challenges remain. The development process for ADCs is notoriously complex. With every reagent, process step and technology transfer step having the potential to introduce impurities or shift critical quality attributes (CQAs), developing a robust methodology is essential.
Key Challenges Associated with ADC Development
As each ADC is unique, obstacles during development may vary significantly between projects. However, a major challenge across the ADC space is management of the highly cytotoxic compounds they contain. These compounds present a significant risk to scientists who come into contact with them, and as such, implementing clear safety protocols and handling procedures to minimize operator exposure is critical. Additionally, the chemical complexity of ADCs necessitates the selection of optimal components. Understanding how to design and select optimal chemical linkers in particular is essential to developing safe and effective therapies. A linker molecule must be able to ensure ADC stability during preparation, storage, and systemic circulation, but also be able to disengage at the right moment, enabling selective release and precise delivery of the payload to tumor cells. Adhering to regulatory standards throughout ADC development is also of paramount importance. Currently, the U.S. Food and Drug Administration (FDA) classifies ADCs as biologics rather than chemically synthesized entities. As a result, the approval process for ADCs is intricate, and requires proof of chemical similarity to the reference product to ensure clinical significance in safety, purity, and potency.
The Role of CDMOs: Streamlining ADC Innovation

Partnering with an experienced contract development and manufacturing organization (CDMO) can aid ADC development in several respects. CDMOs provide the necessary analytical and technical support for an ADC project to succeed and ultimately enable the full potential of these innovative therapeutics to be realized.
- Simplifying the supply chain
With so many moving parts, ADC development typically involves multiple production facilities and reliable access to a wide range of chemical components. CDMOs can provide extensive knowledge of pharmaceutical supply chain operations, as well as proven technology transfer processes, to make sure that each step adheres to the requisites of development and production.
- Providing regulatory guidance
As an emerging modality, the regulatory demands across ADC development are complex. Forging a robust collaboration with an experienced CDMO can be instrumental in overcoming these challenges, allowing innovators to confidently navigate the regulatory landscape.
- Applying analytical expertise
Ultimately, the success of an ADC project depends on chemical understanding. For any given ADC project, a wide range of instrumentation and specialist advice will be required, from tailored purification processes to advanced biologics processing. CDMOs can provide rapid bespoke method development and comprehensive process characterization, which determines ADC viability during scale up and, eventually, in the therapeutic domain. A seasoned CDMO that is able to offer filtration, chromatography, and formulation expertise supported by a comprehensive suite of analytical methods must therefore be selected.
- Ensuring safety and success
Employing stringent safety measures is paramount due to the potency of the cytotoxic component. The need for advanced analytical capabilities that ensure consistent efficacy and safety in each batch is further underscored by the variability between ADC products in the development pipeline. CDMOs can provide their clients with the insight needed to quickly establish the necessary occupational exposure limits (OELs) and storage protocols, as well as conduct a rigorous risk and containment evaluation.
Supporting ADC Programs with Capacity-Solutions
With a global network and team of highly experienced scientists, Veranova has the technical ability to unlock the potential of ADCs. Our skilled process chemists and analytical scientists possess over a decade of experience in the development and scale up of linker molecules for antibody-drug conjugates (ADCs), including several years of experience in polymer drug conjugates and other drug delivery applications. We empower ADC programs with leading expertise, flexible manufacturing, and seamless commercialization, advancing ADCs from Phase 2 to market with flexible, scalable capacity.
Looking Toward a Bright Future
Possessing the power to target diseased cells while minimizing side effects, ADCs are set to increase in popularity over the coming years. Moreover, as the industry’s chemical understanding expands, the application of ADCs is likely to spill into areas beyond oncology. For example, ADCs are predicted to demonstrate therapeutic potential in the treatment of immunological disorders, kidney failure, and arthritis.3 In order to usher in the next wave of ADC successes, an integrated strategy that incorporates meticulous target selection, chemical fine-tuning, and stringent safety protocols will be essential. This comprehensive approach, enabled by strategic partnerships with expert CDMOs such as Veranova, is paving the way for ADC development and deployment. As these innovative therapeutics gain momentum, the result will be a significant positive impact on the pharmaceutical landscape and patients alike.
References
- Grand View Research. Antibody Drug Conjugates Market Size, Share & Trends Analysis Report By Application (Blood Cancer, Breast Cancer), By Technology (Type-cleavable, Non-cleavable), By Product, By Target, By Region, And Segment Forecasts, 2024 – 2030. 2023. https://www.grandviewresearch.com/industry-analysis/antibody-drug-conjugates-market
- Grairi, M. and Le Borgne, M. Antibody-drug Conjugates: prospects for the next generation. Drug Discovery Today 2024, 29, 12. https://doi.org/10.1016/j.drudis.2024.10424
- Pal, L.B., Bule, P., Khan, W., Chella, N. An Overview of the Development and Preclinical Evaluation of Antibody–Drug Conjugates for Non-Oncological Applications. Pharmaceutics 2023, 15, 1807. https://doi.org/10.3390/pharmaceutics15071807


